Vero Cell Line Clonality Assessment
Vero cells are considered as the most widely accepted continuous cell line by the regulatory authorities for the manufacture of viral vaccines for human
use. Reasons for the extensive use of the Vero cell line are the consistent high viral yields and susceptibility to infect a multiple range of viruses, as
well as relatively easy adaptation for growth in bioreactors on microcarriers, thus allowing up-scaling without loss of cell productivity.
In recent years proof of clonality has become a focus. Many companies have received comments back on this topic during the IND review process of their
biological products. Clonality is thought to minimize the heterogeneity of cell banks and thus allow for consistent manufacture a product. Here, Creative
Bioarray has established a high-throughput clonality analysis (FISH) strategy to fast-track your stable cell line development and accelerate subsequent IND
review.
There are regulatory considerations regarding the clonal derivation of the cell line used to create the master cell bank (MCB). Though no one single
regulatory guidance speaks definitively to the need of having a demonstrably "clonally derived" cell line, multiple guidance documents allude to
this general expectation and underscore it as a basic expectation. These include, but are not limited to:
-
ICH Q5D Derivation and Characterization of Cell Substrates Used for Production of Biotechnological/Biological Products, "For recombinant products,
the cell substrate is the transfected cell containing the desired sequences which has been cloned from a single cell progenitor"
-
US FDA Points to Consider in the Manufacture and Testing of Monoclonal Antibody Products for Human Use notes that "The MCB is defined as a
collection of cells of uniform composition derived from a single tissue or cell", and
-
EMA/CHMP guidance which notes "The cell substrate to be used for the production of the monoclonal antibodies should be a stable and continuous
monoclonal cell line that has been developed by means of recombinant DNA and/or other suitable technologies".
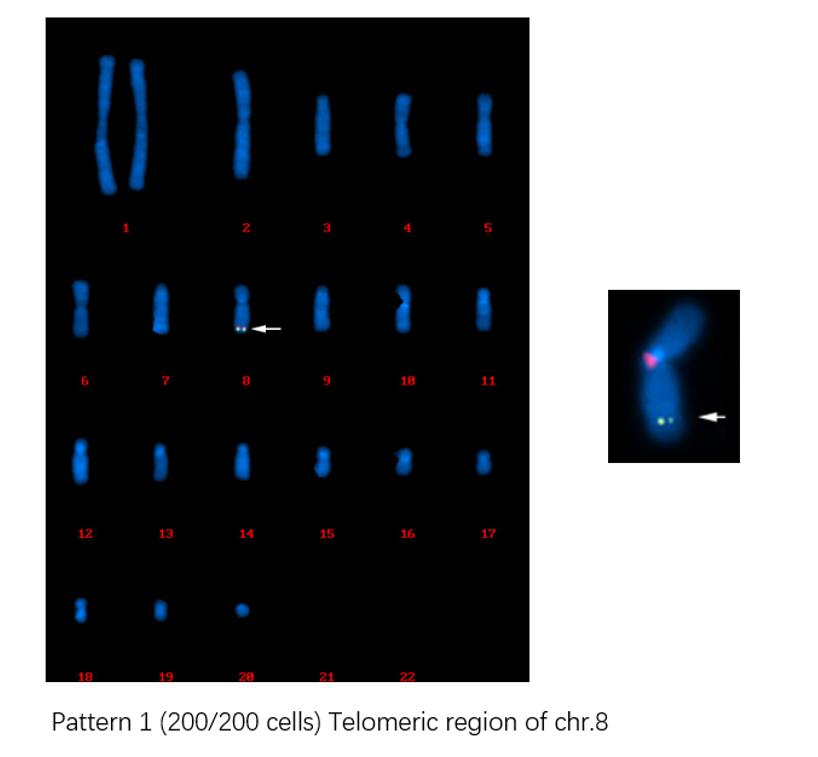
Creative Bioarray's Vero Cell Line Clonality Assessment Service has the following Features:
- Detection of each individual integration site
- Analyze 100-200 cells per sample
- Rapid 4-week turnaround time, including addition reporting
- High accuracy and sensitively
- Competitive pricing
Creative Bioarray offers Vero Cell Line Clonality Assessment Service for your project as follows:
- Probe design
- Probe synthesis
- Chromosome spread preparation
- FISH on chromosome spreads Imaging
- Data analysis (karyotyping and integration pattern analysis)
Quotation and ordering
Our customer service representatives are available 24hr a day! We thank you for considering Creative Bioarray as your Vero Cell Line Clonality Assessment
Service partner.
References
-
Frye, Christopher, et al. "Industry view on the relative importance of "clonality" of biopharmaceutical-producing cell lines."
Biologicals 44.2 (2016): 117-122.
- Wurm, Florian M., and Maria João Wurm. "Cloning of CHO cells, productivity and genetic stability—a discussion." Processes 5.2 (2017): 20.
- ICH Q5D Derivation and characterisation of cell substrates used for production of biotechnological/biological products (CPMP/ICH/294/95)
-
Welch, J. (2017). Tilting at clones: A regulatory perspective on the importance of "Clonality" of mammalian cell banks. CDER/OPQ/OBP/DBRRIV
April 24, 2017.
-
Paul Wu, et al. "Tools and methods for providing assurance of clonality for legacy cell lines" in "Cell Culture Engineering XVI", A.
Robinson, PhD, Tulane University R. Venkat, PhD, MedImmune E. Schaefer, ScD, J&J Janssen Eds, ECI Symposium Series, (2018).
-
Wu P, Hartman T, Almond L, Stevens J, Thrift J, Ojha J, Alves C, Shaw D, Laird MW, Emmins R, Zhu Y, Liu R, Du Z, Koehler R, Jostock T, Anderson K,
Campbell C, Clarke H. Advancing Biologics Development Programs with Legacy Cell Lines: Advantages and Limitations of Genetic Testing for Addressing
Clonality Concerns Prior to Availability of Late Stage Process and Product Consistency Data. PDA J Pharm Sci Technol. 2020 Mar-Apr;74(2):264-274. doi:
10.5731/pdajpst.2018.009316. Epub 2019 Sep 13. PMID: 31519780.
All products and services on this website are only suitable for non-medical purposes.






