ASO and ncRNA
Creative Bioarray provides a comprehensive non-coding RNA visualization analysis solution. Our service is suitable for the research of gene silencing in the development of therapeutic methods and the biological application of disease pathology research. Use the newly advanced RNA-FISH technology and imaging methods to locate and image related transcripts. At the same time, we also provide a combined IHC and other protein in-situ visual analysis program to realize the multi-level analysis of transcription and translation.
ASO and RNAi
The evidence that non-coding sequences are involved in the regulation of gene expression is becoming more and more comprehensive. In recent years, gene silencing therapy has gradually emerged. Methods to inhibit mRNA expression include the use of single-stranded antisense oligodeoxynucleotides (ASO) and RNA interference (RNAi) using double-stranded short interfering RNA molecules (siRNA). The method of using synthetic oligonucleotides to degrade cellular RNA has been used as a method to study gene function. The ASO used for therapeutic intervention is a single-stranded, chemically modified oligonucleotide with a length of 14 to 20 nucleotides, which has the ability to target genes encoding proteins associated with human diseases. The binding of ASO to the target mRNA results in the spatial inhibition of translation by the ribosomal complex. siRNA targets a specific endogenous RNA sequence by guiding the complex, and then the target RNA transcript is combined and degraded by the endonuclease activity of RISC.
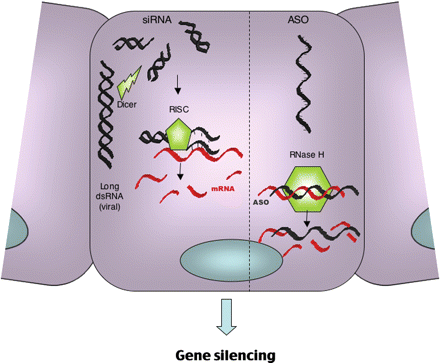 Fig 1. The mechanism of action of ASO versus RNA interference. (Wang J H, et al. 2007)
Fig 1. The mechanism of action of ASO versus RNA interference. (Wang J H, et al. 2007)
Visual Analysis of Non-coding Small RNA and ASO
The method of using synthetic oligonucleotides to degrade cellular RNA has been used as a method to study gene function. At the same time, more and more studies have discovered the role of miRNA, siRNA and other sRNAs in the pathophysiology of diseases. The medical application of gene silencing and the research on the function of ncRNA have promoted the increasing demand for single-cell in situ analysis. Our service aims to provide RNA in situ analysis solutions based on highly specific probes. These uniquely designed commercial probe products have highly specific targeting and can be used to understand the expression patterns and functions of non-coding small RNAs. We use commercialized probe products to complete analysis and testing services. The service process includes service negotiation, probe selection or customization, in situ hybridization and imaging, and data analysis.
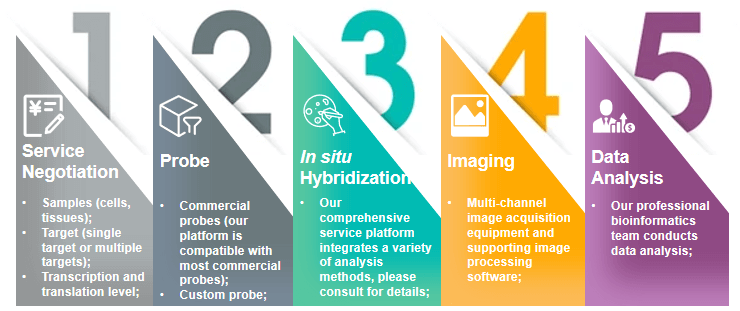 Fig 2. Our ASO and non-coding RNA in situ analysis solutions.
Fig 2. Our ASO and non-coding RNA in situ analysis solutions.
Technical Advantages
- Compared with methods such as NGS, which can lead to the loss of spatial information and a partial understanding of organizational structure and function, in-situ visual analysis helps researchers expand their horizons and provide more intuitive "evidence".
- Realize the analysis of spatial expression patterns at single cell resolution.
- Combine with IHC to realize multi-level expression analysis (transcription, translation).
- Highly specific probes achieve high sensitivity and specificity by amplifying target-specific signals.
- Visualize gene expression and gene regulation with the morphological background.
Applications in biology and medicine
Assess the therapeutic effect, biodistribution and mechanism analysis of ASO and small RNA in gene silencing therapy.
lncRNA, siRNA, miRNA tissue or cell location analysis and functional research.
Research on the development of drug targets related to neurodegenerative diseases through gene silencing.
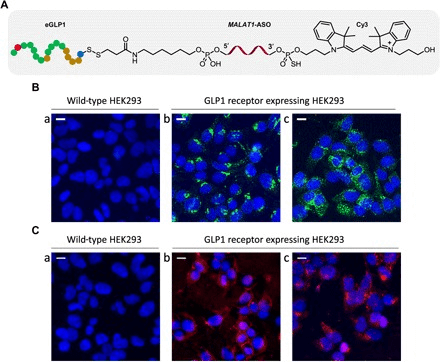 Fig 3. A case of ASO in-situ analysis. (Ämmälä C, et al. 2018)
Fig 3. A case of ASO in-situ analysis. (Ämmälä C, et al. 2018)
If you are interested in our FISH service using probes constructed by the BAC method, please contact us for cooperation. We look forward to cooperating with you in the near future.
References
- Wang J H, Hendry B M, Sharpe C C. Silencing genes in the kidney: antisense or RNA interference?[J]. 2008.
- Ämmälä C, Drury W J, Knerr L, et al. Targeted delivery of antisense oligonucleotides to pancreatic β-cells[J]. Science advances, 2018, 4(10): eaat3386.
All products and services on this website are only suitable for non-medical purposes.


 Fig 1. The mechanism of action of ASO versus RNA interference. (Wang J H, et al. 2007)
Fig 1. The mechanism of action of ASO versus RNA interference. (Wang J H, et al. 2007) Fig 2. Our ASO and non-coding RNA in situ analysis solutions.
Fig 2. Our ASO and non-coding RNA in situ analysis solutions. Fig 3. A case of ASO in-situ analysis. (Ämmälä C, et al. 2018)
Fig 3. A case of ASO in-situ analysis. (Ämmälä C, et al. 2018)


