CONTACT SUPPORT
Online Inquiry
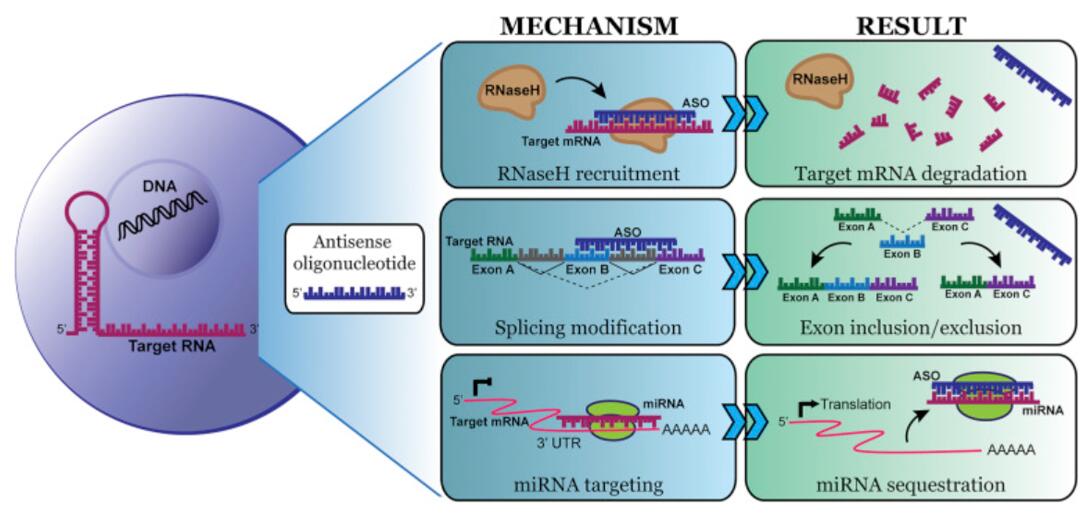
The RNA ISH assay from Creative Bioarray can visualize and monitor the delivery and biodistribution of small interfering RNAs (siRNAs) and anti-sense oligonucleotides (ASOs) molecules to access the specificity and efficacy of these therapies.
In the context of siRNA/ASO gene therapy, RNA ISH can be used to detect and quantify the delivery and uptake of siRNA molecules into target cells. RNA ISH probes specific to the siRNA/ASO molecules can be designed, allowing for their visualization and localization in the cells. ISH can provide valuable information regarding the efficiency and specificity of siRNA/ASO delivery and uptake. By analyzing the presence and distribution of the labeled siRNA/ASO molecules in the cells, researchers can determine the extent of cellular uptake and the level of intracellular siRNA/ASO localization. RNA ISH assay can help optimize the design and delivery methods of siRNA gene therapy, ensuring effective and targeted delivery of therapeutic siRNA molecules.
Synthetic oligonucleotides such as siRNAs and ASOs can target specific genes and regulate their expression. Over the last three decades, siRNA and ASO have merged as powerful tools to modulate the expression of genes in vivo and in vitro. Potential oligonucleotide therapeutics have been discovered and developed with mixed success. For example, in recent years, Mipomersen, Inotersen, Patisiran, and Givlaari have all been approved for use. Mipomersen, a lipid-lowering agent that targets the ApoB gene, was eventually approved by the United States Food and Drug Administration (FDA) in 2013 and later discontinued due to adverse effects.
However, developing oligonucleotide therapeutics remains challenging as they often have unintended off-target effects and cause non-specific toxicity. Additionally, the main hurdle for the application to a broader range of disorders is delivery to target tissues. The recent FDA approval of Patisiran represents a first-of-its-kind RNA interference (RNAi) therapeutic for the treatment of the polyneuropathy of hereditary transthyretin-mediated (hATTR) amyloidosis in adults and the first-ever FDA approval of a siRNA treatment.
Our siRNA/ASO Biodistribution services can help you validate and accelerate your gene therapy development. We have supported dozens of cell and gene therapy companies for research studies enabling pre-IND submission.
siRNA/ASO Biodistribution studies are performed to determine the in vivo distribution of a siRNA/ASO product support early biotherapeutic drug development. This assessment is thus an essential part of the overall safety profile of the gene therapy product. Existing studies are generally limited to organ-level resolution without examining other routes of administration. The siRNA/ASO Biodistribution (RNA ISH assay) Services from Creative Bioarray provide an unparalleled sensitive and specific method for cell- and tissue-specific assessment of siRNA/ASO and cell marker expression analysis in any tissue.
Our customer service representatives are available 24hr a day! We thank you for choosing Creative Bioarray as your preferred siRNA/ASO Biodistribution Service provider.
References