HEK293 Cell Bank Characterization
Recombinant proteins such as hormones, growth factors, cytokines, and monoclonal antibodies play an important role in modern medicine, being used to treat a
variety of disease. Mammalian cell expression systems are the ideal choice to produce eukaryotic proteins as they enable post-translational modifications
which are crucial for the correct folding of many eukaryotic proteins. Human embryonic kidney 293 (HEK293) cells are the predominant hosts
for expression of recombinant proteins and are used for stable expression of proteins.
Creative Bioaray ChromoGenetics has extensive experience in characterization of cGMP HEK293 cell banks used in production of biologic drugs. Our HEK293 cell
line characterization allows you to efficiently streamline your cGMP-compliant manufacturing processes and accelerate toward successful commercialization.
Furthermore, our reporting fulfils FDA and EMA regulatory requirements for pharmaceuticals for human use (ICH guidelines Q5B and Q5D, CTD Quality module,
section 3.2.S.2.3).
HEK293 Cell Line Characterization Tests Available:
HEK293 Cell Bank Characterization
Cell Line Scope
According to document ICH Q5D ‘Quality of Biological Products: Derivatives and Characterization of Cellular Substrates for the Production of
Biotechnology/Biological Products’, Creative Bioarray performs HEK293 cell bank characterization on the following cell lines:
- Research Cell Bank (RCB)
- Working Cell Bank (WCB)
- Master Cell Bank (MCB)
- End-of-Production Cells (EOPC)
Cell Bank characterization is an essential step in ensuring the safety of biopharmaceutical products with the aim of confirming the identity, purity and
genetic stability of cell lines. This characterization can ensure that the cell banks are free of contaminants and are able to provide a solid, stable
foundation to continue production process years down the road. International regulatory guidelines such as ICH (Q5A, Q5B, Q5D), EP and USP for cell lines
characterization mainly focus on four areas:
- The origin and history of the cell line cellular morphology and growth characteristics
- Purity of the cell lines (i.e., absence of contaminating cells, microbial contaminations, and contaminations by adventitious viruses)
- Tumorigenicity and oncogenicity
- Genetic Stability
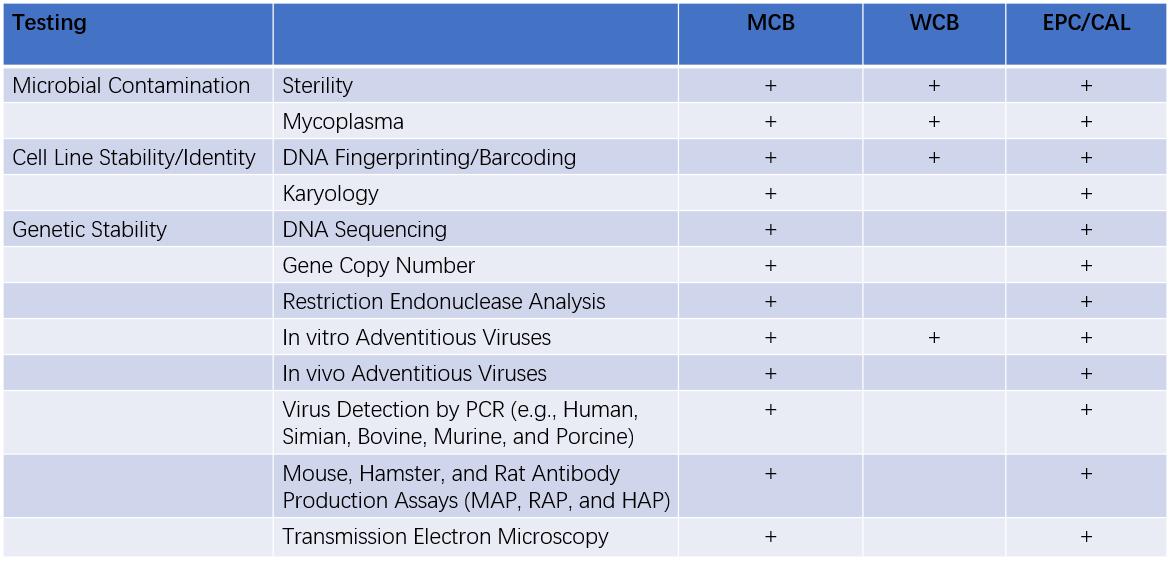
Mammalian Cell Line Characterization- Recommended Testing Plan
Quotation and ordering
Our customer service representatives are available 24 hours a day! We thank you for considering Creative Bioarray as your HEK293 Cell Bank Characterization Service partner.
References
-
Plavsic, Mark. "Q5D derivation and characterization of cell substrates used for production of biotechnological/biological products." ICH
Quality Guidelines (2017): 375-393.
- Galbraith, Daniel. "ICH Q5A: Viral Safety of Biotechnology Products." ICH Quality Guidelines: An Implementation Guide (2017): 311-335.
-
Welch, J. (2017). Tilting at clones: A regulatory perspective on the importance of "Clonality" of mammalian cell banks. CDER/OPQ/OBP/DBRRIV
April 24, 2017.
-
Paul Wu, et al. "Tools and methods for providing assurance of clonality for legacy cell lines" in "Cell Culture Engineering XVI", A.
Robinson, PhD, Tulane University R. Venkat, PhD, MedImmune E. Schaefer, ScD, J&J Janssen Eds, ECI Symposium Series, (2018).
-
Frye, Christopher, et al. "Industry view on the relative importance of "clonality" of biopharmaceutical-producing cell lines."
Biologicals 44.2 (2016): 117-122.
-
Dangi, A. K. Cell Line Techniques and Gene Editing Tools for Antibody Production: A Review. Front Pharmacol. 2018; 9: 630. doi:
10.3389/fphar.2018.00630.
All products and services on this website are only suitable for non-medical purposes.






