Naked DNA Biodistribution
Naked DNA Biodistribution (RNA ISH assay) Services from Creative Bioarray provide an unparalleled sensitive and specific method for the safety and efficiency of gene therapy vector and transgene expression analysis in any tissue.
Increases in efficiency have made naked DNA gene transfer a viable method for gene therapy. RNA ISH assay has several advantages in the application of naked DNA biodistribution gene therapy studies which include:
- Localization: RNA ISH assay allows for the localization of specific mRNA molecules and the plasmid DNA at both the cellular and subcellular levels within a tissue context. This can help in determining the exact location where the delivered gene is being expressed.
- Quantification: RNA ISH assay allows for the quantification of the transcript level of genes of interest. This can facilitate the measurement of gene expression levels after the delivery of naked DNA.
- Visualization: RNA ISH assay produces highly specific and bright signals which can be spatially resolved. This can help in visualizing the biodistribution of the delivered gene within the treated individual or model organism.
- Heterogeneity: RNA ISH assay can detect cellular heterogeneity in gene expression within a population of cells. This can be useful in assessing the efficiency and specificity of naked DNA delivery techniques.
- Temporal information: RNA ISH assay can capture dynamic changes in gene expression levels over time providing a temporal resolution that could be highly valuable in the context of gene therapy studies.
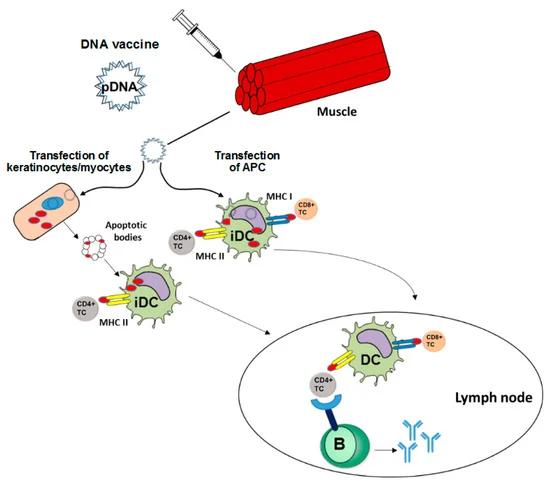 Figure 1. DNA vaccines induce adaptive immune responses.
Figure 1. DNA vaccines induce adaptive immune responses.
RNA ISH assay provides the detection of the expression of the transferred genes through RNA ISH indirectly provides information about the distribution and the efficacy of the naked DNA-based gene transfer.
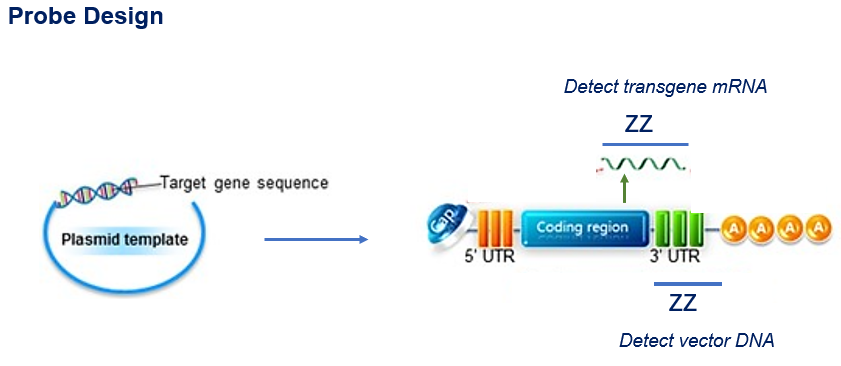 Figure 2. RNA ISH probe design strategy.
Figure 2. RNA ISH probe design strategy.
The transfer of naked DNA for gene therapy is gaining increasing attention for various gene therapy applications and has advantages in terms of ease of DNA preparation, simplicity of use, and lack of immunogenicity. Given its potential in successfully delivering therapeutic genes into targeted cells and tissues with little adverse effects, naked DNA holds the promise of treating a wide range of diseases, from cancer and genetic diseases to infectious diseases. Furthermore, with continuous research and development in delivery techniques and DNA formulation, the efficacy and specificity of Naked DNA gene therapy are expected to be significantly improved in the future, making it a growing field with great potential in medicine. Our Naked DNA Biodistribution services can help you accelerate your gene therapy development.
Naked DNA Biodistribution Tests Available:
Naked DNA Biodistribution
Quotation and ordering
Our customer service representatives are available 24hr a day! We thank you for choosing Creative Bioarray as your preferred Naked DNA Biodistribution Service provider.
References
- Prazeres, D.M.F.; Monteiro, G.A. Plasmid Biopharmaceuticals. Microbiol. Spectrum 2014, 2.
- Fioretti, D.; Iurescia, S.; Rinaldi, M. Recent advances in design of immunogenic and effective naked DNA vaccines against cancer. Recent Pat. Anti-Canc. Drug Discov. 2014, 9, 66–82
- Scheiblhofer, S.; Thalhamer, J.; Weiss, R. DNA and mRNA vaccination against allergies. Pediat. Allergy Immu. 2018.
- Zhang, N.; Nandakumar, K.S. Recent advances in the development of vaccines for chronic inflammatory autoimmune diseases. Vaccine 2018, 36, 3208–3220.
- Maslow, J.N. Vaccines for emerging infectious diseases: Lessons from MERS coronavirus and Zika virus. Hum. Vacc. Immunother. 2017, 13, 2918–2930.
- Weniger, B.G.; Anglin, I.E.; Tong, T.; Pensiero, M.; Pullen, J.K. Workshop report: Nucleic acid delivery devices for HIV vaccines: Workshop proceedings, National Institute of Allergy and Infectious Diseases, Bethesda, Maryland, USA, May 21, 2015. Vaccine 2018, 36, 427–437.
- Tiptiri-Kourpeti, A.; Spyridopoulou, K.; Pappa, A.; Chlichlia, K. DNA vaccines to attack cancer: Strategies for improving immunogenicity and efficacy. Pharmacol. Therapeut. 2016, 165, 32–49.
- Marino, M.; Scuderi, F.; Provenzano, C.; Bartoccioni, E. Skeletal muscle cells: From local inflammatory response to active immunity. Gene Ther. 2011, 18, 109–116.
- Hengge, U.R.; Chan, E.F.; Foster, R.A.; Walker, P.S.; Vogel, J.C. Cytokine gene expression in epidermis with biological effects following injection of naked DNA. Nat. Genet. 1995, 10, 161–166.
- Porgador, A.; Irvine, K.R.; Iwasaki, A.; Barber, B.H.; Restifo, N.P.; Germain, R.N. Predominant role for directly transfected dendritic cells in antigen presentation to CD8+ T cells after gene gun immunization. J. Exp. Med. 1998, 188, 1075–1082.
- Bai, H.; Lester, G.M.S.; Petishnok, L.C.; Dean, D.A. Cytoplasmic transport and nuclear import of plasmid DNA. Biosci. Rep. 2017, 37.
All products and services on this website are only suitable for non-medical purposes.
 Figure 1. DNA vaccines induce adaptive immune responses.
Figure 1. DNA vaccines induce adaptive immune responses. Figure 2. RNA ISH probe design strategy.
Figure 2. RNA ISH probe design strategy.