mRNA/Biomarker Protein Biodistribution Analysis
The chemistry of nucleic acids has experienced a great development since the deciphering of the human genome in 2003. This advance has enabled the identification of new biological targets, responsible for numerous pathologies. mRNA has shown therapeutic potential in a wide range of applications, including viral vaccines, protein replacement therapies, cancer immunotherapies, as well as genome reprogramming and editing. Over the past ten years, these RNA and DNA-based therapies have been developed. Creative Bioarray offers the RNA ISH and IHC assay to visualize and monitor the delivery and biodistribution of these synthetic mRNA molecules and related proteins to access the specificity and efficacy of these therapies. Our mRNA/Biomarker Protein Biodistribution Analysis services can help you validate and accelerate your gene therapy development.
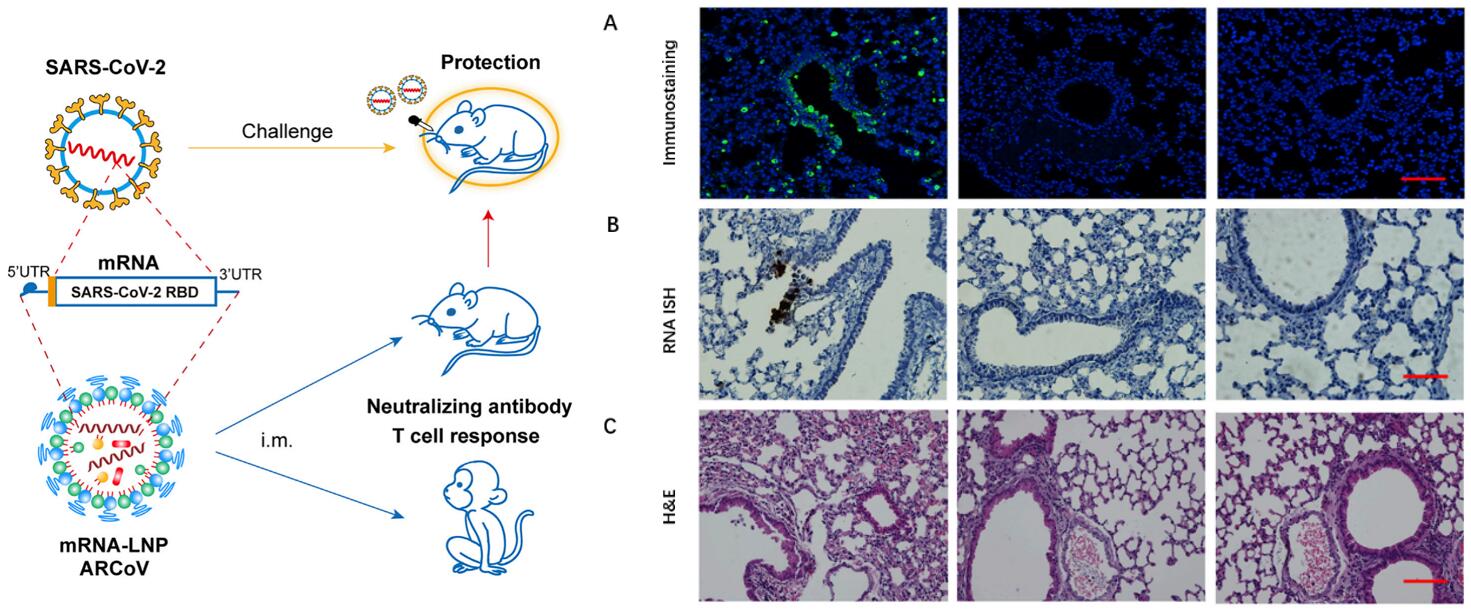 Figure 1. Protection of ARCoV against SARS-CoV-2 Challenge in Mice. Forty days after the initial immunization, mice were inoculated i.n. with the mouse-adapted SARS-CoV-2 (MASCp6), and the indicated tissues were collected 5 days after challenge for detection of viral loads and lung pathology. (A) Immunostaining of lung tissues with a SARS-CoV-2 S-specific mAb. Scale bar, 100 mm. (B) ISH assay for SARS-CoV-2 RNA. Scale bar, 50 mm. Positive signals are shown in brown. (C) H&E staining of lung pathology. Scale bar, 100 mm. Representative images from 4 or 5 mice are shown.
Figure 1. Protection of ARCoV against SARS-CoV-2 Challenge in Mice. Forty days after the initial immunization, mice were inoculated i.n. with the mouse-adapted SARS-CoV-2 (MASCp6), and the indicated tissues were collected 5 days after challenge for detection of viral loads and lung pathology. (A) Immunostaining of lung tissues with a SARS-CoV-2 S-specific mAb. Scale bar, 100 mm. (B) ISH assay for SARS-CoV-2 RNA. Scale bar, 50 mm. Positive signals are shown in brown. (C) H&E staining of lung pathology. Scale bar, 100 mm. Representative images from 4 or 5 mice are shown.
RNA in situ hybridization (RNA FISH) assay combines Biomarker protein immunohistochemistry (IHC) assay offer several advantages and applications in the development process of RNA vaccines.
- Accuracy and Visualization: RNA ISH assay allows for visualizing the spatial distribution of specific mRNA molecules at subcellular resolution in cells and tissues. This is critical in biodistribution analysis for showing where the vaccine is actually going inside the body. Furthermore, protein IHC allows to see the presence and location of the generated target protein, which is the intended product of the administered RNA vaccine.
- Quantification: Both RNA ISH and protein IHC methods enable quantitative measurement. RNA ISH quantifies the amount of target mRNA in each examined cell, while protein IHC can be used to quantify the level of target protein. These quantifications can assist in vaccine dose optimization.
- High Sensitivity: RNA FISH offers high sensitivity, enabling the detection of even low abundance mRNA molecules. This sensitivity can reveal potentially important details that other less sensitive techniques might miss.
- Versatility: Both RNA FISH and protein IHC are versatile techniques and can be used on various types of cells and tissues. This is particularly useful when studying the action of RNA vaccines in different cell types or tissues.
- Verification: Protein IHC can be used to verify the production of the desired protein from the mRNA vaccine.
Thus, employing RNA FISH and protein IHC methods for biodistribution analysis can be valuable for RNA vaccine development, particularly for understanding and optimizing vaccine effectiveness and safety.
Features of mRNA/Biomarker Protein Biodistribution Analysis
(1) Detect and identify cellular subtypes
(2) RNA molecule analysis
(3) Protein analysis
(3) Custom probes designed within 1-2 weeks
(4) Fastest turnaround time
Benefits of mRNA/Biomarker Protein Biodistribution Analysis
(1) Detect and identify cellular subtypes
(2) Visualize gene regulation with morphological context
(3) Validate protein biomarkers in intact tissues
(4) Assess small RNA therapeutic delivery mechanism
(5) Evaluate biodistribution and efficacy of therapy
(6) Add a visual dimension to heterogeneous tissue biology and analysis
Creative Bioarray offers mRNA/Biomarker Protein Biodistribution Analysis for you as follows:
Probe design
Probe synthesis
ISH staining
IHC staining
Imaging
Data analysis
Quotation and ordering
Our customer service representatives are available 24hr a day! We thank you for considering Creative Bioarray as your mRNA/Biomarker Protein Biodistribution Analysis partner.
References
- Sahin, U., Karikó, K. & Türeci, Ö. mRNA-based therapeutics — developing a new class of drugs. Nat. Rev. Drug Discov. 13, 759–780 (2014).
- Pardi, N., Hogan, M. J., Porter, F. W. & Weissman, D. mRNA vaccines — a new era in vaccinology. Nat. Rev. Drug Discov. 17, 261–279 (2018).
- Hajj, K. A. & Whitehead, K. A. Tools for translation: non-viral materials for therapeutic mRNA delivery. Nat. Rev. Mater. 2, 17056 (2017).
- Xiong, Q., Lee, G. Y., Ding, J., Li, W. & Shi, J. Biomedical applications of mRNA nanomedicine. Nano Res. 11, 5281–5309 (2018).
- Li, B., Zhang, X. & Dong, Y. Nanoscale platforms for messenger RNA delivery. Wiley Interdiscip. Rev. Nanomed. Nanobiotechnol. 11, e1530 (2019).
- Meng, C., Chen, Z., Li, G., Welte, T. & Shen, H. Nanoplatforms for mRNA therapeutics. Adv. Ther. 4, 2000099 (2021).
- Kim, J., Eygeris, Y., Gupta, M. & Sahay, G. Self-assembled mRNA vaccines. Adv. Drug Deliv. Rev. 170, 83–112 (2021).
- Baden, L. R. et al. Efficacy and safety of the mRNA-1273 SARS-CoV-2 vaccine. N. Engl. J. Med. 384, 403–416 (2021).
- Anderson, E. J. et al. Safety and immunogenicity of SARS-CoV-2 mRNA-1273 vaccine in older adults. N. Engl. J. Med. 383, 2427–2438 (2020).
- Alameh, Mohamad-Gabriel, Drew Weissman, and Norbert Pardi. "Messenger RNA-based vaccines against infectious diseases." mRNA Vaccines (2020): 111-145.
- Bahl, Kapil, et al. "Preclinical and clinical demonstration of immunogenicity by mRNA vaccines against H10N8 and H7N9 influenza viruses." Molecular Therapy 25.6 (2017): 1316-1327.
- Becerra‐Flores, Manuel, and Timothy Cardozo. "SARS‐CoV‐2 viral spike G614 mutation exhibits higher case fatality rate." International journal of clinical practice 74.8 (2020): e13525.
All products and services on this website are only suitable for non-medical purposes.
 Figure 1. Protection of ARCoV against SARS-CoV-2 Challenge in Mice. Forty days after the initial immunization, mice were inoculated i.n. with the mouse-adapted SARS-CoV-2 (MASCp6), and the indicated tissues were collected 5 days after challenge for detection of viral loads and lung pathology. (A) Immunostaining of lung tissues with a SARS-CoV-2 S-specific mAb. Scale bar, 100 mm. (B) ISH assay for SARS-CoV-2 RNA. Scale bar, 50 mm. Positive signals are shown in brown. (C) H&E staining of lung pathology. Scale bar, 100 mm. Representative images from 4 or 5 mice are shown.
Figure 1. Protection of ARCoV against SARS-CoV-2 Challenge in Mice. Forty days after the initial immunization, mice were inoculated i.n. with the mouse-adapted SARS-CoV-2 (MASCp6), and the indicated tissues were collected 5 days after challenge for detection of viral loads and lung pathology. (A) Immunostaining of lung tissues with a SARS-CoV-2 S-specific mAb. Scale bar, 100 mm. (B) ISH assay for SARS-CoV-2 RNA. Scale bar, 50 mm. Positive signals are shown in brown. (C) H&E staining of lung pathology. Scale bar, 100 mm. Representative images from 4 or 5 mice are shown.