CONTACT SUPPORT
Online Inquiry
oligonucleotide therapeutics (OTs) that regulate gene expression have been developed towards clinical use at a steady pace for several decades, especially siRNA and ASO, but in recent years the field has been accelerating. To date, there are 11 marketed products based on antisense oligonucleotides, aptamers and small interfering RNAs, and many others are in the pipeline for both academia and industry.
However, developing OTs remains challenging as they often have unintended off-target effects and cause non-specific toxicity. Additionally, the efficient delivery of OTs to target the specific cells is critical to the successful development of OTs.
Therefore, biodistribution study plays a significant role in tracking these molecules. In Situ Hybridization is an excellent method to detect the siRNA/ASOs, as many current ways are either not sensitive enough to detect low levels of expression or incapable of detecting siRNA+ or ASO+ cells in the tissue context. RNA ISH combines IHC which allows the analysis of siRNA/ASO, target mRNA, and biomarker protein (e.g., cytokines, tissue-specific proteins, or target proteins), respectively, while preserving the cellular relationship and tissue architecture. Researchers have found that combining or multiplexing both technologies provide unique information. siRNA/ASO/Biomarker Protein Biodistribution Analysis service from Creative Bioarray can help validate and accelerate your gene therapy development.
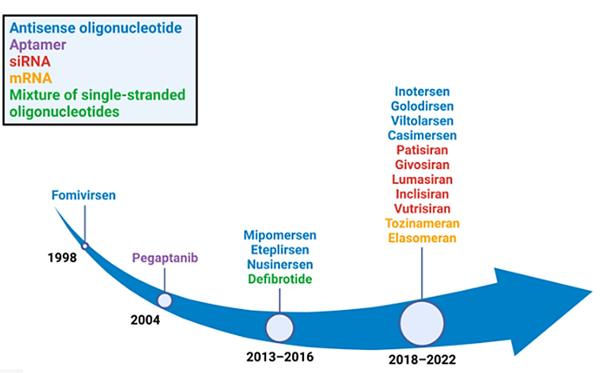 Figure 1. Years in which RNA therapeutics gained FDA approval. Eight antisense oligonucleotide drugs (blue), five single interfering RNA drugs (red), two mRNA drugs (orange), one aptamer drug (purple), and a mixture of single-stranded oligonucleotide drugs (green) were approved.
Figure 1. Years in which RNA therapeutics gained FDA approval. Eight antisense oligonucleotide drugs (blue), five single interfering RNA drugs (red), two mRNA drugs (orange), one aptamer drug (purple), and a mixture of single-stranded oligonucleotide drugs (green) were approved.
(1) Detection and identify cellular subtypes
(2) Synthetic RNA molecule Biomarker analysis
(3) Biomarker protein analysis
(4) Custom probes designed within 1-2 weeks
(5) Fastest turnaround time
(6) Experienced scientists for accurate data analysis
(7) Standard and customized experiment plan
(1) Simultaneously visualize in vivo delivery of siRNA/ASP and biomarker protein (e.g., cytokines, tissue specific proteins or target proteins)
(2) Visualize gene regulation and protein expression with morphological context
(3) Validate siRNA/ASO in intact tissues
(4) Evaluate biodistribution and efficacy of therapy
(5) Assess small RNA therapeutic delivery mechanism
(6) Add a visual dimension to heterogeneous tissue biology and analysis
Our customer service representatives are available 24hr a day! We thank you for considering Creative Bioarray as your siRNA/ASO/Biomarker Protein Biodistribution Analysis partner.
References