Preimplantation Genetic Diagnosis
Preimplantation genetic diagnosis (PGD) was developed in the late 1980s as an alternative to prenatal diagnosis for couples at risk of passing genetic or chromosomal abnormalities to their children. As an alternative to prenatal diagnosis, genetic testing of biopsied cells from embryos produced in vitro is used for couples who are at risk of passing genetic or chromosomal abnormalities to their children. The technology has been used as an embryo selection tool for IVF patients, screening as many chromosomes as possible, in the hope that selecting chromosomally normal embryos will lead to higher implantation rates and lower miscarriage rates. PGD is a procedure used to detect genetic defects in preimplantation embryos to avoid their transfer to the uterus and ensure pregnancy from genetically normal embryos. Embryos diagnosed as abnormal during a PGD cycle can be used to establish hESC lines with genetic diseases, PG screening (PGS) for chromosomal abnormalities and single-gene disease diagnosis for detection of genetic mutations. These PGD-affected embryos are not intended for transfer or cryopreservation, but they can serve as a valuable source for deriving hESC lines. The use of hESCs and induced pluripotent stem cells in disease-specific studies, and the application of stem cell technology in drug discovery and toxicity testing is discussed.
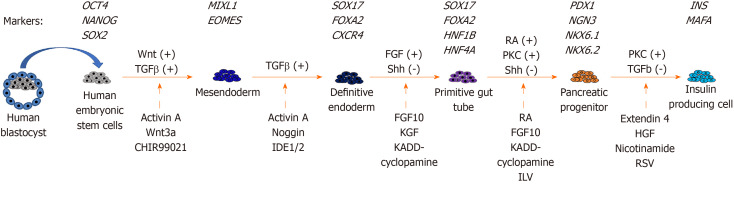 Fig 1. Stepwise in vitro differentiation of pancreatic cells from human embryonic stem cells. (Andy Chun-Hang Chen et al., 2020)
Fig 1. Stepwise in vitro differentiation of pancreatic cells from human embryonic stem cells. (Andy Chun-Hang Chen et al., 2020)
Cytogenetic Analysis of PGD-derived hESCs
FISH technology can be used to establish hESC cell lines with genetic diseases. Within ethically reasonable limits, hESCs derived from abnormal embryos after PGD screening is of great significance for the study of human genetic diseases. In theory, this strategy is also applicable to the development of embryo selection strategies in animal medical research and animal breeding work, and the study of outstanding genetic traits.
Application
Molecular tests for non-diagnostic purposes in studies of in vitro fertilization techniques.
For pre-transplantation genetic analysis research and technological innovation research.
Methods of genetic testing in research projects related to advanced pregnancy.
FISH-based cytogenetic diagnosis can be used in embryos produced by in vitro fertilization (IVF), where cells are biopsied and analyzed using genetic tests such as PCR and FISH.
FISH-based cytogenetic diagnosis can be used to screen for genetic abnormalities commonly found in PGD cases, such as cystic fibrosis and hemophilia, or chromosomal abnormalities, such as translocations.
FISH-based cytogenetic diagnosis can be used to study cell differentiation and developmental biology, as well as other areas of research focused on the potential use of cell therapy for the treatment of degenerative human diseases.
FISH-based cytogenetic diagnosis can be used for the study of in vitro disease models derived from hESC lines derived from embryos with monogenic diseases or chromosomal aberrations.
FISH-based cytogenetic diagnosis can help hESC lines study human genetics, gene expression patterns, and understand events that occur during human embryonic development in the presence of genetic disorders.
FISH-based cytogenetic diagnosis can aid PGD-derived cell lines in drug screening and development of new possible treatments.
 Fig 2. FISH assay for genetic diagnosis of PGD-derived hESCs.
Fig 2. FISH assay for genetic diagnosis of PGD-derived hESCs.
Creative Bioarray provides cytogenetic diagnosis and chromosome mapping services. We are deeply involved in the application field of FISH technology and provide various types of probe customization and testing services. If you are interested in our service, please contact us for cooperation. We look forward to cooperating with you in the near future.
References
- Chen, Andy Chun-Hang, et al. "Human embryonic stem cells as an in vitro model for studying developmental origins of type 2 diabetes." World Journal of Stem Cells 12.8 (2020): 761.
- Harper J C. Preimplantation genetic screening[J]. Journal of medical screening, 2018, 25(1): 1-5.


 Fig 1. Stepwise in vitro differentiation of pancreatic cells from human embryonic stem cells. (Andy Chun-Hang Chen et al., 2020)
Fig 1. Stepwise in vitro differentiation of pancreatic cells from human embryonic stem cells. (Andy Chun-Hang Chen et al., 2020) Fig 2. FISH assay for genetic diagnosis of PGD-derived hESCs.
Fig 2. FISH assay for genetic diagnosis of PGD-derived hESCs.


