CONTACT SUPPORT
Online Inquiry
The CAR-NK Biodistribution (RNA ISH assay or RNA ISH combine IHC assay) Services from Creative Bioarray provide an unparalleled sensitive and specific method for cell and tissue-specific assessment of CAR-NK (Chimeric antigen receptor-natural killer) therapy vector, cytokines, and transgene expression analysis in any tissue.
Biodistribution studies play a crucial role in the efficient and successful implementation of CAR-NK technology. Understanding the in vivo biodistribution of CAR-NK cells is vital in predicting therapeutic effects, potential off-target toxicities, and overall safety profile. Furthermore, it also helps in predicting the pharmacokinetics and pharmacodynamics of CAR-NK cells that is essential for their clinical efficacy.
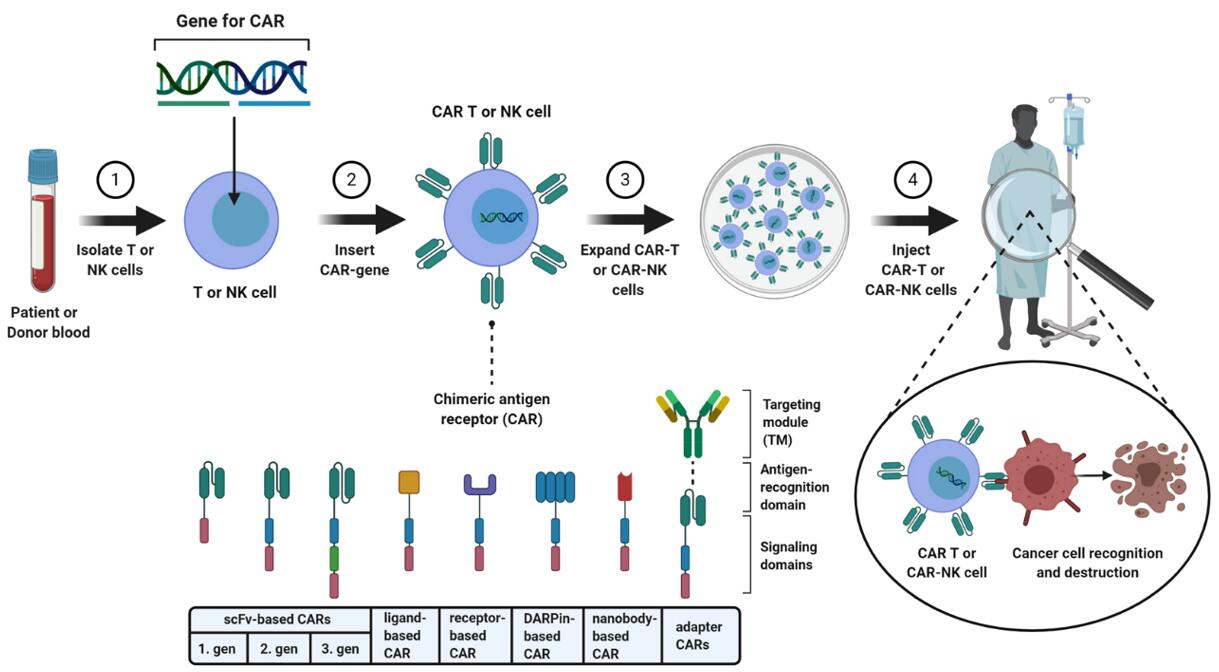 Fig. 1: Schematic illustration of a CAR-T or CAR-NK cell therapy, which uses primary immune cells.
Fig. 1: Schematic illustration of a CAR-T or CAR-NK cell therapy, which uses primary immune cells.
As for the regulatory information, CAR-NK cells fall into the category of genetically modified organisms (GMOs). In the USA, the Food and Drug Administration (FDA) manages their regulation, while in the European Union, it's overseen by the European Medicines Agency (EMA). These regulatory agencies require comprehensive preclinical biodistribution studies for GMOs to ensure the utmost safety and efficacy.
The application of RNA in situ hybridization (RNA ISH) and RNA ISH coupled with immunohistochemistry (IHC) in CAR-NK technology biodistribution hold several advantages. RNA ISH allows one to visualize the precise location of the RNA or DNA sequences within an intact cellular framework, which gives critical insights for biodistribution studies.
On the other hand, RNA ISH+IHC allows for the simultaneous detection of RNA and protein at a single-cell level, providing an even more detailed understanding of the spatial distribution and intra-cellular colocalization of CAR-NK cells. This comprehensive cellular level data can enhance the accuracy of biodistribution analysis and further contribute to devising effective therapeutic strategies.
Our customer service representatives are available 24hr a day! We thank you for choosing Creative Bioarray as your preferred CAR-NK Biodistribution Service provider.
References