CONTACT SUPPORT
Online Inquiry
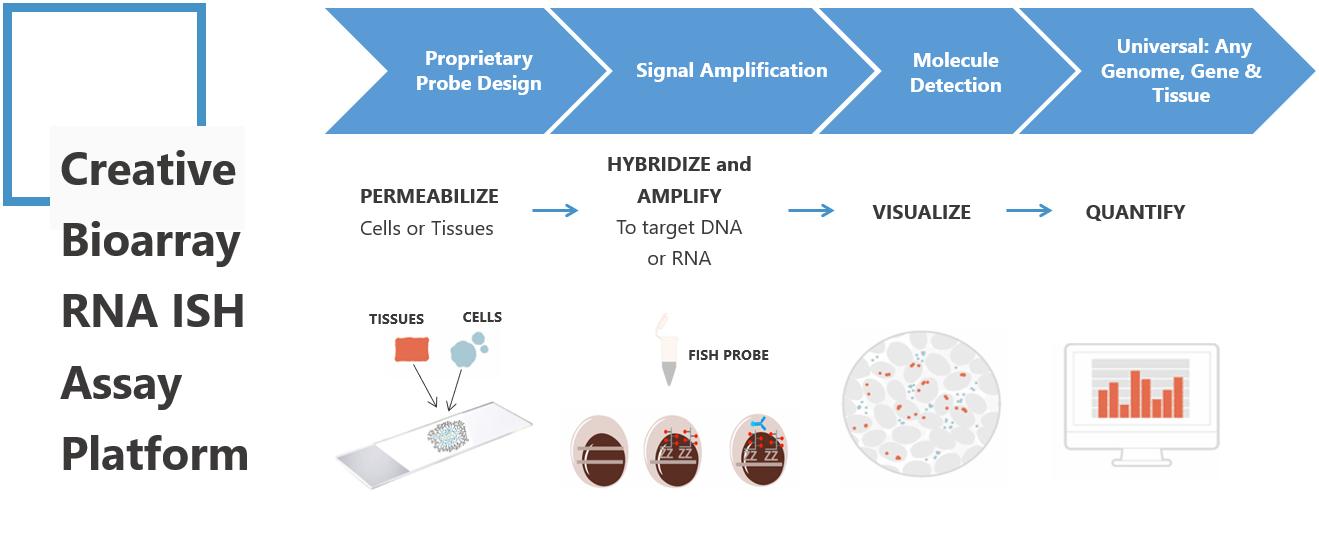
Targeted gene therapy has shown great promise for treating rare diseases, particularly those that arise from single gene defects. Recombinant adeno-associated virus (AAV) has recently emerged as a highly popular gene delivery vector for a wide range of gene therapy and vaccine applications. One essential consideration when evaluating the therapeutic potential and safety profile of an AAV vector is its distribution and persistence in various organs and tissues. While qPCR-based methods provide average values for copy number, the RNA in situ hybridization (ISH) technology provides morphology-based, cell-specific quantification of vector DNA, transgene mRNA, and specific biomarkers.
As stated by the USFDA, "The biodistribution data, coupled with other preclinical safety endpoints such as clinical pathology and histopathology, help determine whether vector presence or gene expression correlates with any tissue-specific detrimental effects in animals." (FDA Guidance for Industry: Preclinical Assessment of Investigational Cellular and Gene Therapy Products, November 2013). This is particularly important in germ-line tissues (i.e., gonads) to determine intertied transmission status. Our AAV Vector/Transgene/Biomarker RNA Biodistribution Analysis service can help you validate and accelerate your gene therapy development.
The RNA ISH technology is an excellent solution for detecting AAV vector DNA and therapeutic transgene mRNA expression within intact tissue morphology, addressing critical questions on tissue biodistribution, persistence, cellular tropism, and vector promoter activity. As a leading technology provider, Creative Bioarray's elite scientists have broad experience in AAV Vector/Transgene/Biomarker RNA Biodistribution Analysis (RNA ISH method), which can supply the best service for you.
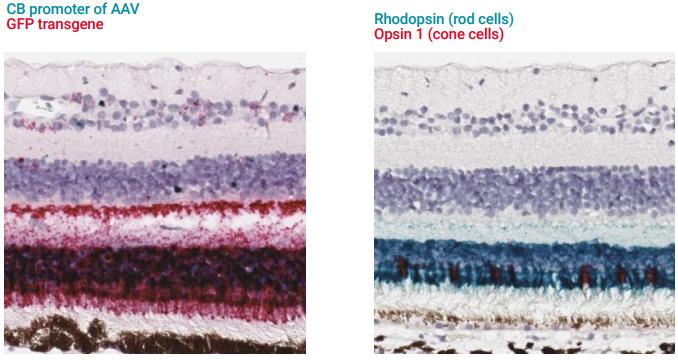 Figure 1. Visualization of AAV-transduced cells and specific cell types in the monkey retina. (A) The AAV vector used in this study contains a GFP transgene under the control of the CB promoter. RNAscope® probes were designed to target the CB promoter DNA sequence (V-CB promoter) as well as the GFP transgene mRNA (GFP-C2). (B) RNAscope® probes were used to detect the rod cells marked by Rhodopsin (Mfa-RHO) and cone cells marked by Opsin 1 short wave sensitive (Mfa-OPN1SW-C2).
Figure 1. Visualization of AAV-transduced cells and specific cell types in the monkey retina. (A) The AAV vector used in this study contains a GFP transgene under the control of the CB promoter. RNAscope® probes were designed to target the CB promoter DNA sequence (V-CB promoter) as well as the GFP transgene mRNA (GFP-C2). (B) RNAscope® probes were used to detect the rod cells marked by Rhodopsin (Mfa-RHO) and cone cells marked by Opsin 1 short wave sensitive (Mfa-OPN1SW-C2).
(1) Detection of single-copy DNA vectors
(2) Codon-optimized transgene and biomarker expression analysis
(3) Custom probes designed within 1-2 weeks
(4) Fastest turnaround time
(1) Simultaneously visualize in vivo delivery of vector DNA and therapeutic transgene mRNA
(2) Identify cell tropism of AAV vector by multiplexing with cell-type markers
(3) Distinguish transgene from endogenous sequences, at the single nucleotide level
(4) Quantify AAV+ cell number and track persistence over time
Our customer service representatives are available 24hr a day! We thank you for considering Creative Bioarray as your AAV Vector/Transgene/Biomarker RNA Biodistribution Analysis partner.
References