CONTACT SUPPORT
Online Inquiry
A messenger RNA (mRNA) can encode a protein leading to desired biological functions, supporting the rationale for developing mRNA-based therapeutics. Creative Bioarray offers the RNA ISH assay to visualize and monitor the delivery and biodistribution of these synthetic mRNA molecules to access the specificity and efficacy of these therapies. Our mRNA/Biomarker RNA Biodistribution Analysis services can help you validate and accelerate your gene therapy development.
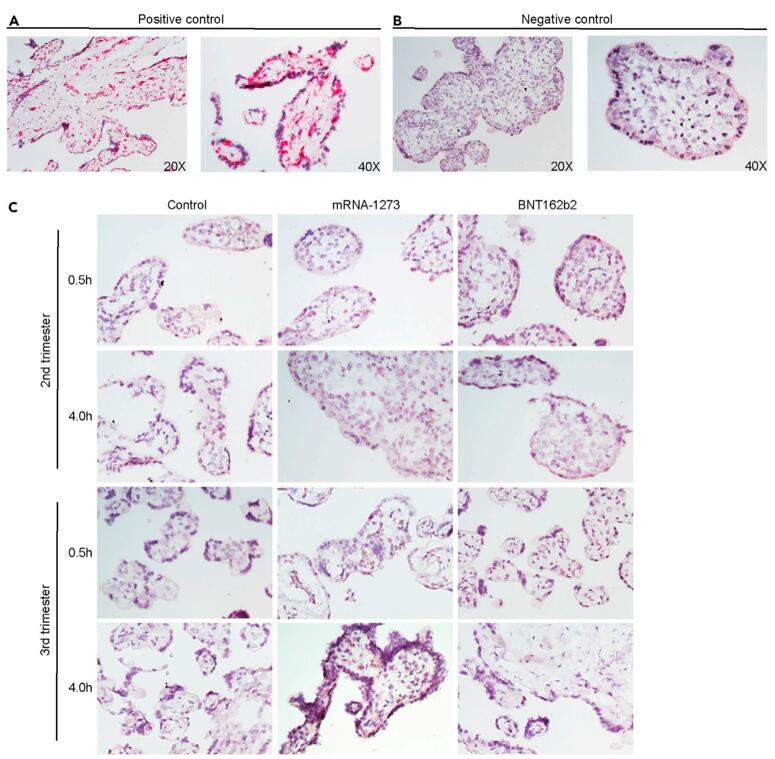 Figure 1. Lack of detection of COVID mRNA vaccine via in situ hybridization in human placental explants. Chorionic villi explants derived from second (n = 2) or third trimester (n = 2) human placentas were incubated with 0.1 mg/mL (not shown) or 1 mg/mL mRNA-1237 or BNT162B2 vaccines. After 0.5 h or 4 h, tissues were fixed, paraffin embedded, sectioned and probed for mRNA vaccine using RNA in situ hybridization (A and B) Positive and (B) negative controls for RNA ISH detection of mRNA vaccine. Peptidylprolyl isomerase B (PPIB) was used as a positive control. Pink dots corresponding to PPIB mRNA can be observed within the chorionic villi at 20X and 40X. DapB was used as a negative control. No signal was detected at 20 or 40X. (C) In situ detection of mRNA vaccine in vaccine-exposed explants. No signal was evident in explants incubated with either vaccine at any of the two time points.
Figure 1. Lack of detection of COVID mRNA vaccine via in situ hybridization in human placental explants. Chorionic villi explants derived from second (n = 2) or third trimester (n = 2) human placentas were incubated with 0.1 mg/mL (not shown) or 1 mg/mL mRNA-1237 or BNT162B2 vaccines. After 0.5 h or 4 h, tissues were fixed, paraffin embedded, sectioned and probed for mRNA vaccine using RNA in situ hybridization (A and B) Positive and (B) negative controls for RNA ISH detection of mRNA vaccine. Peptidylprolyl isomerase B (PPIB) was used as a positive control. Pink dots corresponding to PPIB mRNA can be observed within the chorionic villi at 20X and 40X. DapB was used as a negative control. No signal was detected at 20 or 40X. (C) In situ detection of mRNA vaccine in vaccine-exposed explants. No signal was evident in explants incubated with either vaccine at any of the two time points.
RNA In Situ Hybridization (RNA ISH) assay is a valuable technique in mRNA and Biomarker RNA Biodistribution analysis, especially in the context of RNA vaccine development. Here are some of its advantages and applications:
Through these applications, RNA FISH constitutes an effective tool for mRNA/Biomarker RNA Biodistribution analysis in the process of RNA vaccine development, contributing to both the fundamental mechanistic understanding and translational success of these vaccines.
The field of mRNA vaccines has seen exponential growth in recent times, largely due to the swift approval and widespread distribution of mRNA vaccines targeting COVID-19, which carry the gene for the virus's spike protein. These vaccines are delivered as lipid nanoparticles (LNPs), and their acceptance in clinical settings paves the way for novel therapeutic applications across a multitude of diseases.
(1) Detect and identify cellular subtypes
(2) Synthetic RNA molecule biomarker analysis
(3) Custom probes designed within 1-2 weeks
(4) Fastest turnaround time
(1) Detect and identify cellular subtypes
(2) Visualize gene regulation with morphological context
(3) Validate miRNA biomarkers in intact tissues
(4) Assess small RNA therapeutic delivery mechanism
(5) Evaluate biodistribution and efficacy of therapy
(6) Add a visual dimension to heterogeneous tissue biology and analysis
Our customer service representatives are available 24hr a day! We thank you for considering Creative Bioarray as your mRNA/Biomarker RNA Biodistribution Analysis partner.
References