CONTACT SUPPORT
Online Inquiry
In situ hybridization (ISH) techniques initially developed by Joseph Gall and Mary Lou Pardue in the 1960s (Pardue and Gall 1969) and John et al. (1969) have proved to be powerful tools for determining the chromosomal location of hybridized nucleic acid. In the field of molecular biology, fluorescence in situ hybridization (FISH) techniques have revolutionized our understanding of the organization and function of genetic material within cells. FISH allows us to visualize and map specific DNA or RNA sequences in fixed cells or tissue samples, providing valuable insights into chromosomal abnormalities, gene expression patterns, and spatial organization of genetic material.
FISH techniques are based upon hybridization, with fluorescently labeled RNA or DNA probes that target and bind complementary DNA or RNA sequences in the sample of interest. The probes are made up of nucleotide sequences that are specific to the target sequences, enabling a high level of specificity in binding. Upon binding to its target sequence, the fluorescent label affixed to the probe generates a signal that is detectable via a fluorescence microscope.
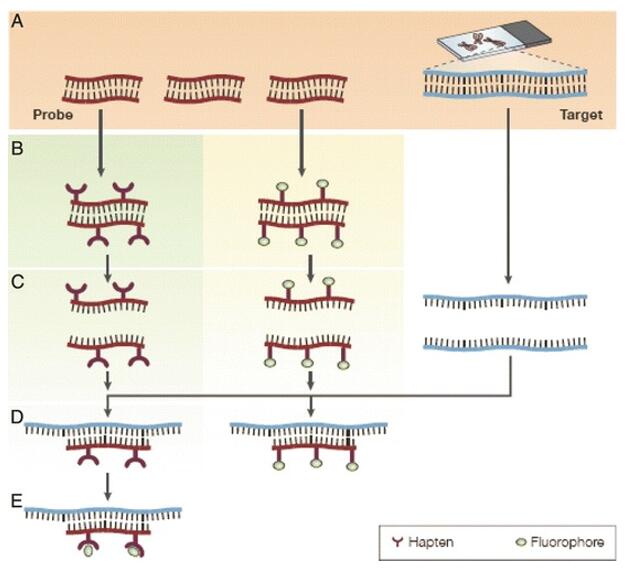 Fig. 1 The principles of fluorescence in situ hybridization. (Shakoori AR, 2017)
Fig. 1 The principles of fluorescence in situ hybridization. (Shakoori AR, 2017)
DNA FISH is a powerful technique used to detect and localize specific DNA sequences within the genome. It has found wide applications in cytogenetics, cancer research, prenatal diagnosis, and gene mapping. In DNA FISH, the DNA probe is typically a short DNA fragment labeled with a fluorescent dye. Scientists use three different types of FISH probes, each of which has a different application.
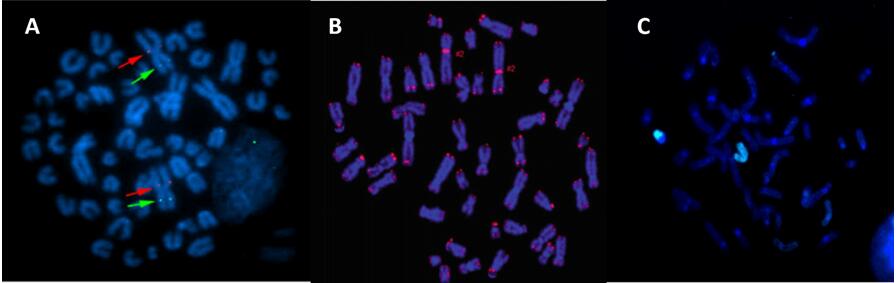 Fig. 2 Sheep-specific gene probe (A), human telomere FISH probe (B), and human chromosome X paint probe (C).
Fig. 2 Sheep-specific gene probe (A), human telomere FISH probe (B), and human chromosome X paint probe (C).
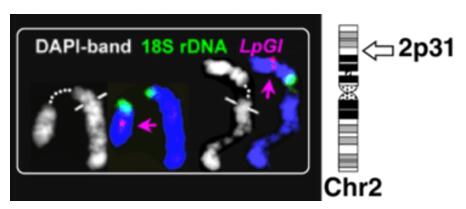 Fig. 3 FISH mapping of LpGI gene. (Ansari HA, et al., 2017)
Fig. 3 FISH mapping of LpGI gene. (Ansari HA, et al., 2017)
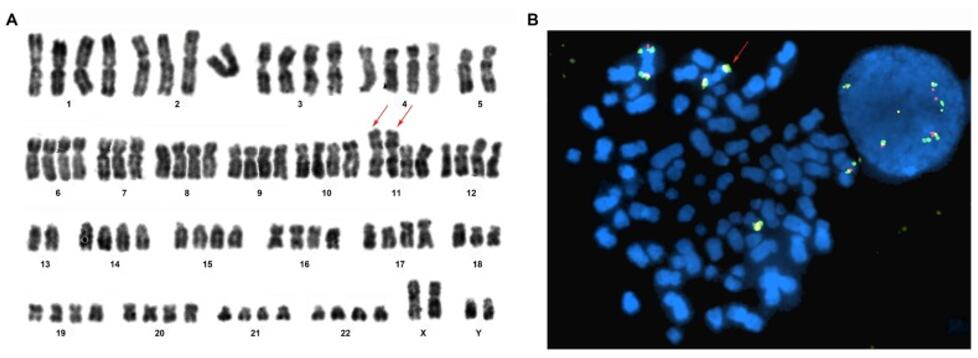 Fig. 4 Karyotype and FISH analysis. (Yang R, et al., 2019)
Fig. 4 Karyotype and FISH analysis. (Yang R, et al., 2019)
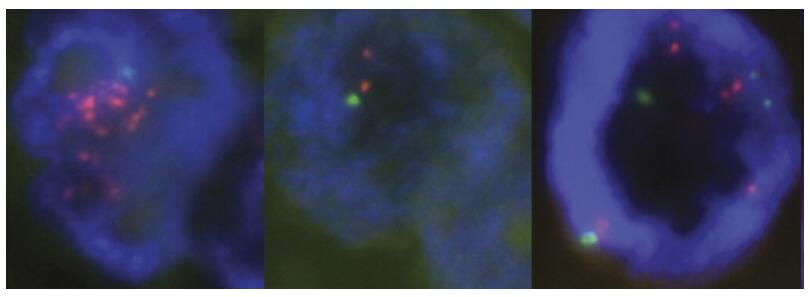 Fig. 5 HER2 gene amplification testing by FISH for diagnosis of breast cancer. (Press MF, et al., 2016)
Fig. 5 HER2 gene amplification testing by FISH for diagnosis of breast cancer. (Press MF, et al., 2016)
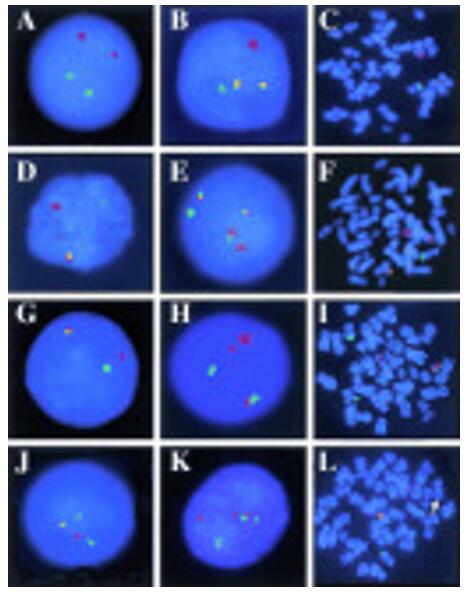 Fig. 6 Detection of double BCR/ABL fusion by FISH in chronic myeloid leukemia. (Dewald GW, et al., 1998)
Fig. 6 Detection of double BCR/ABL fusion by FISH in chronic myeloid leukemia. (Dewald GW, et al., 1998)
While DNA FISH allows the detection of DNA sequences, RNA FISH is specifically designed to visualize and localize RNA molecules within cells. RNA FISH has become an essential tool in studying gene expression, RNA localization, and RNA-protein interactions. It allows researchers to directly observe the spatial and temporal distribution of RNA transcripts at the single-cell level, providing insights into gene expression patterns, RNA localization, and regulation within biological systems.
Launched in 2012, RNAscope is a novel RNA in situ hybridization (ISH) technology that detects target RNAs in tissues and cells. Its proprietary probe is designed to amplify target-specific signals but not background noise from non-specific hybridization. This technique enables the precise detection and spatial localization of RNA expression at single-molecule sensitivity, providing valuable insights into gene expression patterns within the context of intact biological samples.
Compared with traditional RNA in situ hybridization, the specific double Z probe design of RNAscope technology avoids the drawbacks of traditional long-stranded RNA probes and utilizes its own cascade amplification detection principle to detect the target RNAs with high sensitivity. Target RNAs with a specific sequence of greater than or equal to 300 bases can be used for the design of probes. As a result, RNAscope technology can be applied to almost all species, all tissues, and all genes.
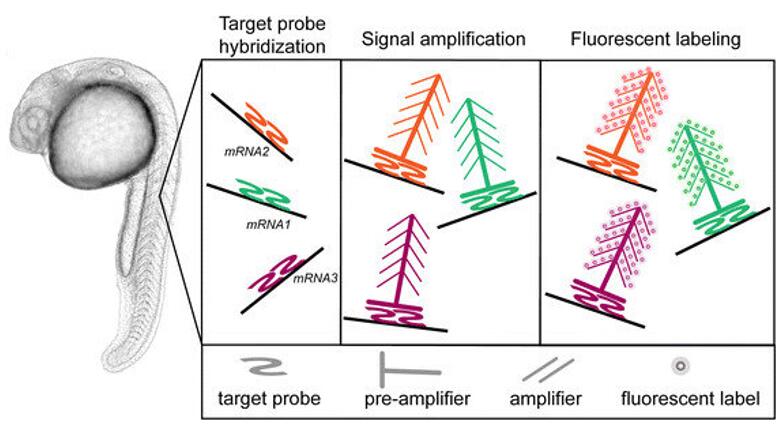 Fig. 7 The RNAscope principle. (Gross-Thebing T, et al., 2014)
Fig. 7 The RNAscope principle. (Gross-Thebing T, et al., 2014)
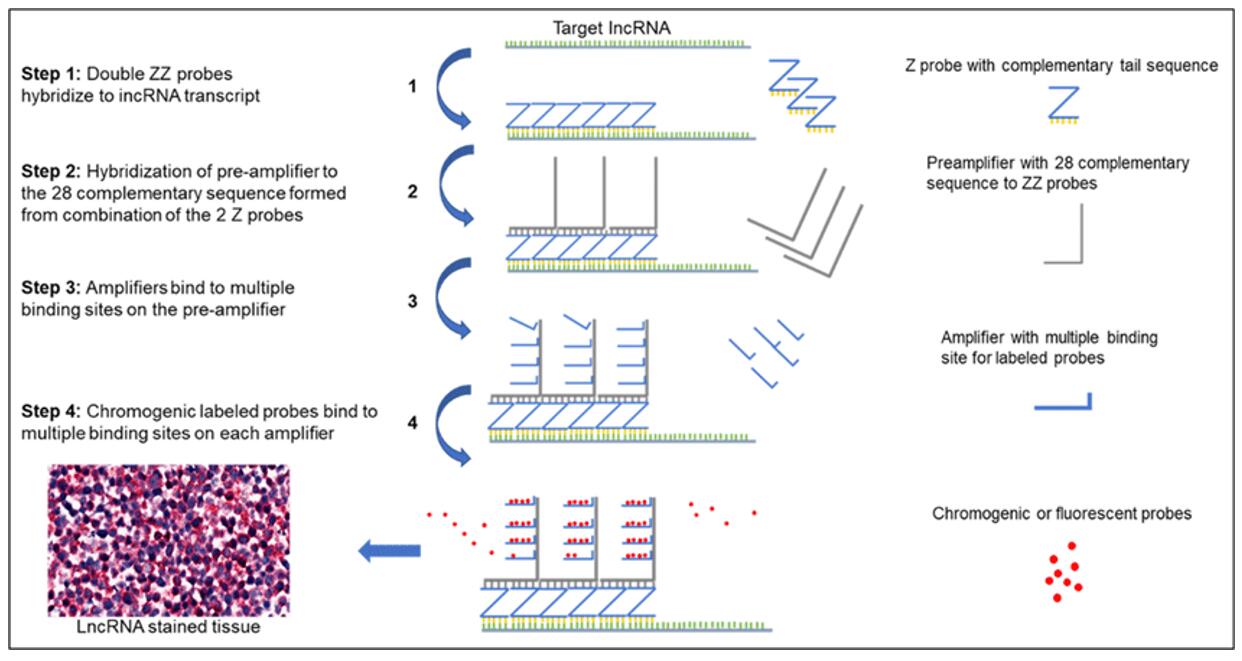 Fig. 8 A schematic representation of Z probe-based RNAScope assay for lncRNA analysis. (Tripathi MK, et al., 2018)
Fig. 8 A schematic representation of Z probe-based RNAScope assay for lncRNA analysis. (Tripathi MK, et al., 2018)
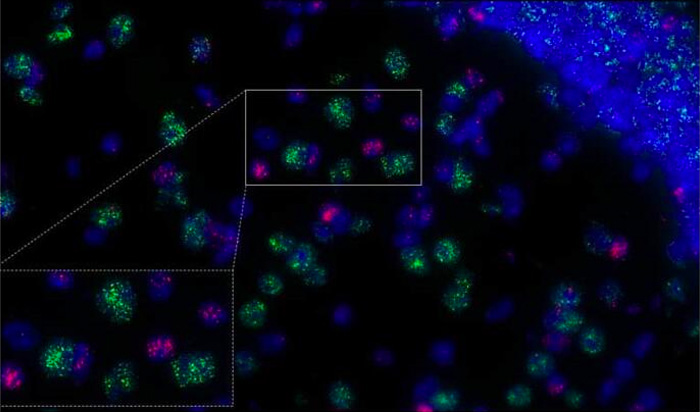 Fig. 9 Detection of lncRNAs in mouse brain hippocampus.
Fig. 9 Detection of lncRNAs in mouse brain hippocampus.
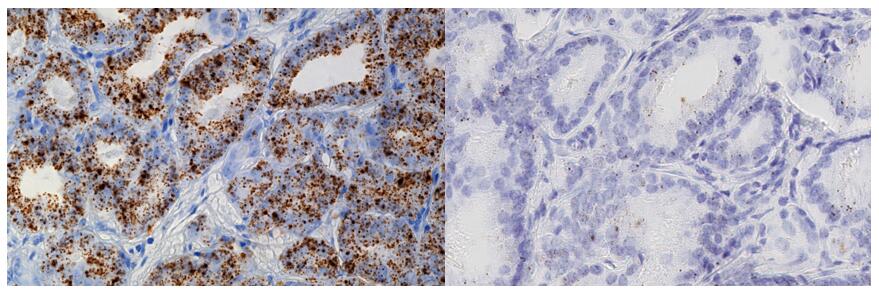 Fig. 10 Evaluation of tissue PCA3 expression in prostate cancer by RNAscope. (Warrick JI, et al., 2014)
Fig. 10 Evaluation of tissue PCA3 expression in prostate cancer by RNAscope. (Warrick JI, et al., 2014)
Although both DNA FISH and RNA FISH utilize similar principles, there are notable differences between the two techniques.
| FISH Techniques | Target | Sample Type | Probe Length | Detection | Multiplex Capability | Applications |
|---|---|---|---|---|---|---|
| DNA FISH | DNA | FFPE cells & tissues (TMAs) Fresh frozen tissues Fixed frozen tissues Cultured cells |
Short DNA fragments, ranging from a few hundred to a few thousand base pairs in length. | Fluorescent | N/A | Cytogenetics, cancer research, prenatal diagnosis, and gene mapping. |
| RNA FISH | RNA | FFPE cells & tissues (TMAs) Fresh frozen tissues Fixed frozen tissues Cultured cells |
RNA fragments, consisting of 20-30 nucleotides. | Fluorescent | N/A | Gene expression analysis, RNA localization, and RNA-protein interactions. |
| RNAscope | mRNA > 300 bases lncRNA > 300 bases |
FFPE cells & tissues (TMAs) Fresh frozen tissues Fixed frozen tissues Cultured cells |
The standard probe design is 20 ZZ probes (minimum of 7 ZZ probes). | Single to up to 12-plex | LncRNA and mRNA >300 bases. | |
| BaseScope Assay | Exon junctions/Splice Variants RNA 50 to 300 bases Validated point mutations |
FFPE cells & tissues (TMAs) Fresh frozen tissues Fixed frozen tissues Cultured cells |
1 to 3 ZZ probes based on the application | Single to duplex | Exon junctions/splice variants, circular RNA, gene fusion, gene knockout Short/highly-homologous sequences, TCRs and CDR sequence for T cell clones, pre-miRNA, gene editing/CRISPR, CAR-T cell validation and detection Point mutation, short InDel, homologues |
|
| miRNAscope Assay | Small RNAs 17-50 bases ASOs, miRNAs, siRNAs |
FFPE cells & tissues (TMAs) Fresh frozen tissues Fixed frozen tissues Cultured cells |
N/A | Single-plex | Small RNAs 17-50 bases ASOs, miRNAs, piRNAs, shRNAs, siRNAs, and tRNAs. |
References